Paper category: Short communication
Corresponding author: Ehsan Atazadeh (atazadeh@tabrizu.ac.ir)
DOI: 10.1515/ohs-2020-0009
Received: 07/06/2019
Accepted: 01/10/2019
Full text: here
Citation (APA style): Dehghani, J., Atazadeh, E., Omidi, Y., et al. (2020). The use of 18S ribosomal DNA, ITS and rbcL molecular markers to study the genus Dunaliella (Dunaliellaceae) in Iranian samples: A phylogenetic approach. Oceanological and Hydrobiological Studies, 49(1), pp. 88-98. Retrieved 10 Mar. 2020, from doi:10.1515/ohs-2020-0009
Abstract
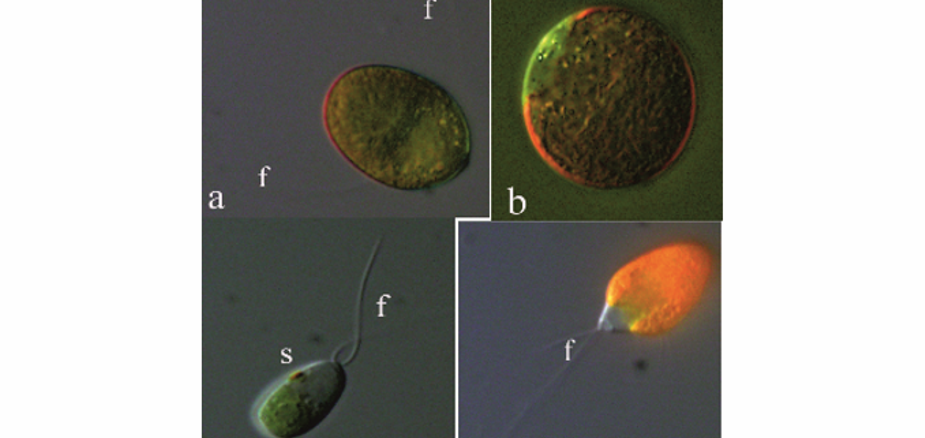
The microalga Dunaliella has been the focus of attention over recent decades owing to its high biotechnological potential for the production of β-carotene, biofuels and even as a good expression system for the production of recombinant proteins. Different species of this genus have unique features, biological characteristics and biotechnological potential. Therefore, it is necessary to have a clear and reliable taxonomic method to identify different species of Dunaliella. Although several taxonomic systems are available for Dunaliella based on morphological, physiological and molecular features, none of these methods are reliable enough and some controversies exist over different classification systems. In the current study, molecular techniques and bioinformatics tools have been used to re-assess the phylogenetic position of Dunaliella species based on 18S ribosomal DNA (18S rDNA), ITS and rbcL regions. The overall findings based on these markers provide a new and more reliable tool for phylogenetic analysis of Dunaliella species/strains.
References
Assuncao, P., Jaen-Molina, R., Caujape-Castells, J., de la Jara, A., Carmona, L. et al. (2012). Molecular taxonomy of Dunaliella (Chlorophyceae), with a special focus on D. salina: ITS2 sequences revisited with an extensive geographical sampling. Aquatic biosystems 8:2. DOI: 10.1186/2046-9063-8-2.
Assunçao, P., Jaen-Molina, R., Caujape-Castells, J., de la Jara, A., Carmona, L. et al. (2012). Phylogenetic position of Dunaliella acidophila (Chlorophyceae) based on ITS and rbcL sequences. J. Appl. Phycol. 24(4): 635–639
Barzegari, A., Hejazi, M.A., Hosseinzadeh, N., Eslami, S., Mehdizadeh Aghdam, E. et al. (2010). Dunaliella as an attractive candidate for molecular farming. Mol. Biol. Rep. 37(7): 3427–3430. DOI: 10.1007/s11033-009-9933-4.
Ben-Amotz, A., Polle, Jr.E. & Subba Rao, D. (2009). The alga Dunaliella. Science Publishers.
Ben-Amotz, A., Katz, A. & Avron, M. (1982). Accumulation of β‐carotene in halotolerant alge: purification and characterization of β‐carotene‐rich globules from Dunaliella bardawil (Chlorophyceae). J. Phycol. 18(4): 529–537.
Borowitzka, M. & Huisman, J. (1993). The ecology of Dunaliella salina (Chlorophyceae, Volvocales): effect of environmental conditions on aplanospore formation. Bot. Mar. 36(3):233–244.
Borowitzka, M.A. & Siva, C.J. (2007). The taxonomy of the genus Dunaliella (Chlorophyta, Dunaliellales) with emphasis on the marine and halophilic species. J. Appl. Phycol. 19(5): 567–590
Cifuentes, A.S., González, M.A., Inostroza, I. & Aguilera, A. (2001). Reappraisal of physiological attributes of nine strains of Dunaliella (Chlorophyceae): growth and pigment content across a salinity gradient. J. Phycol. 37(2): 334–344.
Coleman, A.W., Suarez, A. & Goff, L.J. (1994). Molecular Delineation of species and syngens in volvocacean green algae (Chlorophyta). J. Phycol. 30(1): 80–90.
Dehghani, J., Adibkia, K., Movafeghi, A., Barzegari, A., Pourseif, M.M. et al. (2018). Stable transformation of Spirulina (Arthrospira) platensis: a promising microalga for production of edible vaccines. Applied Microbiology and Biotechnology 102(21): 9267–9278. DOI: 10.1007/s00253-018-9296-7.
Dehghani, J., Movafeghi, A., Barzegari, A. & Barar, J. (2017). Efficient and stable transformation of Dunaliella pseudosalina by 3 strains of Agrobacterium tumefaciens. BioImpacts: BI 7(4): 247–254. DOI: 10.15171/bi.2017.29.
Fredericg, S. & Ramírez, M.E. (1996). Systematic studies of the Antarctic species of the Phyllophoraceae (Gigartinales, Rhodophyta) based on rbcL sequence analysis. Hydrobiologia 326(1): 137–143.
Freshwater, D.W., Fredericq, S., Butler, B.S., Hommersand, M.H. & Chase, M.W. (1994). A gene phylogeny of the red algae (Rhodophyta) based on plastid rbcL. Proc. Natl. Acad. Sci. 91(15): 7281–7285.
Garcia, F., Freile-Pelegrin, Y. & Robledo, D. (2007). Physiological characterization of Dunaliella sp. (Chlorophyta, Volvocales) from Yucatan, Mexico. Bioresource Technology 98(7): 1359–1365. DOI: 10.1016/j.biortech.2006.05.051.
Gomez, P.I., Gonzalez, M.A. & Becerra, J. (1999). Quantity and quality of b-carotene produced by two strains of Dunaliella salina (Teodoresco 1905) from the North of Chile. J. Chil. Chem. Soc. 44 (4): 463–468.
Gonzalez, M.A., Coleman, A.W., Gomez, P.I. & Montoya, R. (2001). Phylogenetic relationship among various strains of Dunaliella (Chlorophyceae) based on nuclear ITS rDNA sequences. J. Phycol. 37(4): 604–611.
Gonzalez, M.A., Gomez, P.I. & Montoya, R. (1998). Comparison of PCR-RFLP analysis of the ITS region with morphological criteria of various strains of Dunaliella. J. Appl. Phycol. 10(6): 573–580.
Hejazi, M.A., Barzegari, A., Gharajeh, N.H. & Hejazi, M.S. (2010). Introduction of a novel 18S rDNA gene arrangement along with distinct ITS region in the saline water microalga Dunaliella. Saline Syst. 6: 4. DOI: 10.1186/1746-1448-6-4.
Johnson, M.K., Johnson, E.J., MacElroy, R.D., Speer, H.L. & Bruff, B.S. (1968). Effects of salts on the halophilic alga Dunaliella viridis. J. Bacteriol. 95(4): 1461–1468.
Kumar, S., Nei, M., Dudley, J. & Tamura, K. (2008). MEGA: a biologist-centric software for evolutionary analysis of DNA and protein sequences. Brief Bioinform. 9(4): 299–306. DOI: 10.1093/bib/bbn017.
Lemieux, C., Vincent, A.T., Labarre, A., Otis, C. & Turmel, M. (2015). Chloroplast phylogenomic analysis of chlorophyte green algae identifies a novel lineage sister to the Sphaeropleales (Chlorophyceae). BMC Evolutionary Biology 15: 264. DOI: 10.1186/s12862-015-0544-5.
Mai, J.C. & Coleman, A.W. (1997). The internal transcribed spacer 2 exhibits a common secondary structure in green algae and flowering plants. J. Mol. Evol. 44(3): 258–271.
Markovits, A., Gianelli, M., Conejeros, R. & Erazo, S. (1993). Strain selection for β-carotene production by Dunaliella. World J. Microbiol. Biotechnol. 9(5): 534–537.
Massyuk, N. (1973). Morphology, taxonomy, ecology and geographic distribution of the genus Dunaliella Teod. and prospects for its potential utilization. Naukova Dumka, Kiev 312.
Melkonian, M. & Preisig, H.R. (1984). An ultrastructural comparison between Spermatozopsis and Dunaliella (Chlorophyceae). Plant Syst. Evol. 146 (1–2): 31–46.
Olmos, J., Ochoa, L., Paniagua-Michel, J. & Contreras, R. (2009). DNA fingerprinting differentiation between β-carotene hyperproducer strains of Dunaliella from around the world. Saline Syst. 5(1): 5.
Olmos, J., Paniagua, J. & Contreras, R. (2000). Molecular identification of Dunaliella sp. utilizing the 18S rDNA gene. Lett. Appl. Microbiol. 30(1): 80–84.
Olsen, G.J., Lane, D.J., Giovannoni, S.J., Pace, N.R. & Stahl, D.A. (1986). Microbial ecology and evolution: a ribosomal RNA approach. Annu. Rev. Microbiol. 40(1): 337–365.
Oren, A. (2005). A hundred years of Dunaliella research: 1905–2005. Saline Syst. 1(1): 2.
Oren, A. (2010). The Alga Dunaliella. Biodiversity, Physiology, Genomics and Biotechnology. J. Phycol. 46(1): 216–217.
Parra, O.O., Floyd, L.G. & Wilcox, L.W. (1990). Taxonomic identification and ultrastructural characterization of a Chilean strain of Dunaliella. Rev. Chil. Hist. Nat.
Proschold, T., Marin, B., Schlosser, U.G. & Melkonian, M. (2001). Molecular phylogeny and taxonomic revision of Chlamydomonas (Chlorophyta). I. Emendation of Chlamydomonas Ehrenberg and Chloromonas Gobi, and description of Oogamochlamys gen. nov. and Lobochlamys gen. nov. Protist 152(4): 265–300. DOI: 10.1078/1434-4610-00068.
Raja, R., Anbazhagan, C., Lakshmi, D. & Rengasamy, R. (2004). Nutritional studies on Dunaliella salina (Volvocales, Chlorophyta) under laboratory conditions. Seaweed Res. Utili. 26: 127–146.
Riisgard, H., Nielsen, K.N., Sogaard-Jensen, B. (1980). Further studies on volume regulation and effects of copper in relation to pH and EDTA in the naked marine flagellate Dunaliella marina. Marine Biol. 56(4): 267–276
Wilcox, L.W., Lewis, L., Fuerst, P. & Floyd, G. (1992). Group I introns within the nuclear-encoded small-subunit rRNA gene of three green algae. Mol. Biol. Evol. 9(6): 1103–1118.

Bądź pierwszy, który skomentuje ten wpis