Paper category: Original research paper
Corresponding author: Rafał P. Ronowski (rafal.ronowski@phdstud.ug.edu.pl)
DOI: 10.1515/ohs-2020-0015
Received: 24/06/2019
Accepted: 26/11/2019
Full text: here
Citation (APA style): Oceanological and Hydrobiological Studies, Volume 49, Issue 2, Pages 157–167, eISSN 1897-3191, ISSN 1730-413X, DOI: https://doi.org/10.1515/ohs-2020-0015.
Abstract
A number of small, shallow inland lakes in the Northern Hemisphere are acidic, soft-water water bodies, mostly fed by precipitation. Some of these lakes are home to Lobelia dortmanna and other aquatic macrophytes from the isoetid group. The present study identifies a plant species replacement trend along the main environmental gradient in such lakes, i.e. pH of water.
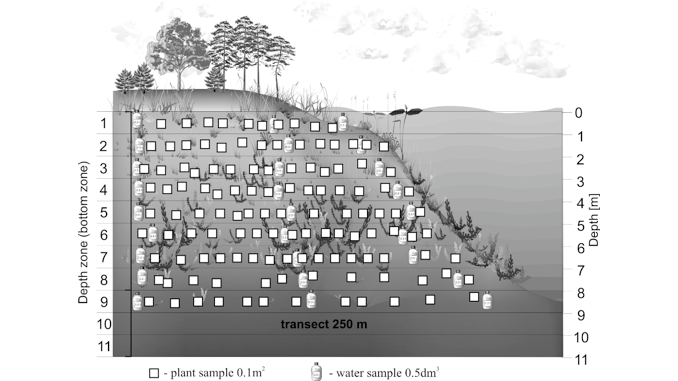
In 70 lakes covered by the study and ordered along the gradient of increasing alkalinity, bryophytes gradually decline. Their decreasing frequency is 86.8% (pH 4.0), 32.5% (pH 6.5) and < 10% (pH 6.6–9.5). At the same time, the frequency of vascular plants increases from 13.2% in strongly acidic lakes up to 92.8% in alkaline ones. The majority of isoetids usually occur at pH 5.5–6.7, but one of them (Littorella uniflora) often (frequency 40.5%) occurs also in eutrophic, alkaline lakes (pH 9.0–9.5), where it forms large populations. In lakes ranked according to the decreasing alkalinity gradient, bryophyte populations have been found to be smaller, while vascular plants become more abundant. The decreasing water acidity leads to partial or total elimination of bryophytes, which are replaced by eutrophilic vascular plants, frequently accompanied by some stonewort species.
References
Arts, G.H.P. (2002). Deterioration of atlantic soft water macrophyte communities by acidification, eutrophication and alcalisation. Aquatic Bot. 73: 373–393.
Banaś, K., Gos, K. & Szmeja, J. (2012). Factors controlling vegetation structure in peatland lakes – Two conceptual models of plant zonation. Aquatic Bot. 96: 42–47.
Banaś, K. (2016). The principal regulators of vegetation structure in lakes of north-west Poland. A new approach to the assembly of macrophyte communities. Wydawnictwo Uniwersytetu Gdańskiego, Gdańsk.
Bociąg, K., Robionek, A., Rekowska, E. & Banaś, K. (2013). Effect of hydrodynamic disturbances on the biomass and architecture of the freshwater macroalga Chara globularis Thuill. Acta Bot. Gallica 160(2): 149–156. DOI: 10.1080/12538078.2013.822826.
Boston, H.L. (1986). A discussion of the adaptations for carbon acquisition in relation to the growth strategy of aquatic isoetids. Aquat. Bot. 26: 259–270.
Boston, H.L., Adams, M.S. & Pienkowski, T.P. (1987). Models of the use of root-zone CO2 by selected North American isoetids. Ann. of Botany 60: 495–503.
Bowes, G. (1987). Aquatic plant photosynthesis. Strategies that enhance carbon gain. In R.M.M. Grawford (Ed.), Plant Life in Aquatic and Amphibious Habitats (pp. 79–98). Blackwell Scientific Publications, Boston.
Chmara, R., Szmeja, J. & Ulrich, W. (2013). Patterns of abundance and co-occurrence in submerged plants communities. Ecological Research 28(3): 387–395.
Chmara, R., Szmeja, J. & Banaś, K. (2014). Factors controlling the frequency and biomass of submerged vegetation in outwash lakes supplied with surface water or groundwater. Boreal Environment Research 19(3): 168–180.
Chmara, R., Banaś, K. & Szmeja, J. (2015). Changes in the structural and functional diversity of macrophyte communities along an acidity gradient in softwater lakes. Flora 216: 57–64.
Feldmann, T. (2012). The structuring role of lake conditions for aquatic macrophytes. Unpublished doctoral dissertation. Tartu. Institute of Agricultural and Environ-mental Sciences, Estonian University of Life Sciences.
Frost-Christensen, H. & Sand-Jensen, K. (1995). Comparative kinetics of photosynthesis in floating and submerged Potamogeton leaves. Aquatic Botany 51: 121–134.
Gittins, R. (1985). Canonical analysis. A review with applications in ecology. Springer-Verlag, Berlin.
Gos, K. & Banaś, K. (1999). Succession trends of submerged vegetation in Pomeranian acidic lakes (NW Poland). Acta Hydrobiol. 41: 171–178.
Hultén, E. & Fries, M. (1986). Atlas of north European vascular plants. Koeltz Scientific Books, Königstein.
IUCN Red List (2015). Retrieved from www.iucn.org
Jalas, J. & Suominen, J. (Eds.) (1972–1992). Atlas Florae Europaeae. The Comm. for Mapping the Flora of Europe and Soc. Biol. Fennica Vanamo. Helsinki.
Kolada, A., Piotrowicz, R., Wilk-Woźniak, E., Dynowski, P. & Klimaszyk, P. (2017). Conservation status of the Natura 2000 habitat 3110 in Poland: Monitoring, classification and trends. Limnol. Rev. 17(4): 215–222.
Mäemets, A. (1974). On Estonian lake types and main trends of evolution. In Estonian Wetland and their life (pp. 29–63). Acad. of Sciences of Estonian SSSR
Madsen, T.V. (1985). A community of submerged aquatic CAM plants in Lake Kalgaard, Denmark. Aquat. Bot. 23: 97–108.
Madsen, T.V. & Sand-Jensen, K. (1991). Photosynthetic carbon assimilation in aquatic macrophytes. Aquatic Botany 41: 5–40.
Madsen, T.V. & Maberly, S.C. (1991). Diurnal variation in light and carbon limitation of photosynthesis by two species of submerged freshwater macrophyte with a differential ability to use bicarbonate. Freshwater Biology 26: 175–187.
Mitchell, S.F. & Perrow, M.R. (1998). Interactions between grazing birds and macrophytes. In E. Jeppesen, M. Søndergaard, M. Søndergaard, K. & Christoffersen K. (Eds.), The Structuring Role of Submerged Macrophytes in Lakes (pp. 175–195) Springer-Verlag, New York.
Murphy, K.J. (2002). Plant communities and plant diversity in softwater lakes of northern Europe. Aquatic Bot. 73: 287–324.
Pulido, C., Sand-Jensen, K., Lucassen, E.C.H.E.T., Roelofs, J.G.M., Brodersen K.P. et al. (2011). Improved prediction of vegetation composition in NW European softwater lakes by combining location, water and sediment chemistry. Aquatic Sciences 74: 351–360.
Rørslett, B. (1991). Principal determinants of aquatic macrophyte richness in northern European lakes. Aquat. Bot. 39: 173–193.
Sand-Jensen, K. (1978). Metabolic adaptation and vertical zonation of Littorella uniflora (L.) Aschers. and Isoëtes lacustris L. Aquatic Bot. 4: 1–10.
Sand-Jensen, K. & Borum, J. (1984). Epiphyte shading and its effect on photosynthesis and diel metabolism of Lobelia dortmanna L. the spring bloom in a Danish lake. Aquat. Bot. 20: 109–119.
Seddon, B. (1965). Occurrence of Isoëtes echinospora in eutrophic lakes in Wales Ecology 46: 747–748.
Seddon, B. (1972). Aquatic macrophytes as limnological indicators. Freshwater Biol. 2: 107–130.
Siraj, S., Yousuf, A.R. & Parveen, M. (2011). Spatio-temporal dynamics of macrophytes in relation to ecology of a Kashmir Himalayan Wetland. Int. Res. J. Biochem. Bioinformatics 1(4): 84–88.
Sokal, R.R. & Rohlf, F.J. (2012). Biometry: the principles and practice of statistics in biological research. W.H. Freeman and Co., 4th edition, New York, 937 pp,
Szmeja, J. (1992). Structure, spatial organization and demography of isoetid populations. Ecological studies of submerged aquatic plants. Gdansk Univ. Press. Ser. Monogr. 75: 1–137. (In Polish).
Szmeja, J. (1996). Register of Polish Lobelia lakes. Fragm. Flor. Geobot. 3: 347–367. (In Polish).
Szmeja, J., Banaś, K. & Bociąg, K. (1997). Ecological conditions and tolerance limits of isoetids along the southern baltic coast. Ekol. Pol. 45(2): 343–359.
Szmeja, J. & Bociąg, K. (2004). The disintegration of populations of underwater plants in soft water lakes enriched with acidic organic matter. Acta Soc. Bot. Pol. 73: 165–173.
Szmeja, J. (2006). A guide to the study of aquatic plants. Wyd. UG, Gdansk, 467 pp. (In Polish).
Szmeja, J. (2010). Changes in the aquatic moss Sphagnum denticulatum Brid. population abundance in a softwater lake over a period of three years. Acta Soc. Bot. Pol. 79(2): 167–173.
Szmeja, J., Bociąg, K. & Merdalski, M. (2010). Effect of light competition with filamentous algae on the population dynamics development of the moss species Warnstorfia exannulata in a softwater lake. Pol. J. Ecol. 58(2): 221–230.
Ter Braak, C.J.F. (1986). Canonical correspondence analysis: a new eigenvector technique for multivariate direct gradient analysis. Ecology 67: 1167–1179.
Ter Braak, C.J.F. (1987). The analysis of vegetation-environmentrelationships by canonical correspondence analysis. Vegetatio 64: 69–77.
Ter Braak, C.J.F. (1988). CANOCO – an extension of DECORANA to analyse species-environment relationships. Vegetatio 75: 159–160.
Ter Braak, C.J.F. & Šmilauer, P. (2002). CANOCO reference manual and CanoDraw for Windows user’s guide: software for Canonical Community Ordination (version 4.5). Microcomputer Power, New York.
Tison, J.-M. & De Foucault, B. (Eds.) (2014). Flora Gallica. Flore de France. Societé Botanique de France. Biotope, Mèze, 1196 pp.
Wetzel, R.G. (1990). Land-water interfaces: Metabolic and limnological regulators. Verhandlungen des Internationalen Verein Limnologie 24: 6–24.
Vestergaard O.S. & Sand-Jensen K (2000). Alkalinity and trophic state regulate aquatic plant distribution in Danish lakes. Aquatic Bot. 67: 85–107. DOI: 10.1016/S0304-3770(00)00086-3.
Vöge, M. (1992). Tauchuntersuchungen an der submersen Vegetation in 13 Seen Deutschlands unter besondener Berücksichtigung der Isoetiden-Vegetation. Limnologica 22(11): 82–96.

Bądź pierwszy, który skomentuje ten wpis